Call us now
07971891546( PIN:252)
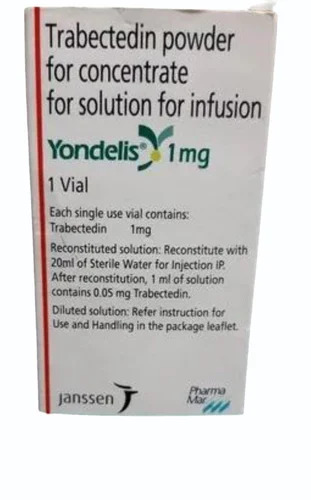

Price:
Price 12500 INR / Bottle
Minimum Order Quantity : 1 Bottle
Dosage Form : Tablet
Salt Composition : Erdafitinib
Shelf Life : 24 months
Indication : Bladder Cancer, Lung Cancer
Price 950 INR / Bottle
Minimum Order Quantity : 10 Bottles
Dosage Form : Tablet
Salt Composition : Exemestane
Shelf Life : 36 months
Indication : Breast Cancer
Price 95000 INR / Vial
Minimum Order Quantity : 1 Vial
Dosage Form : Injection
Salt Composition : Eculizumab
Shelf Life : 36 months
Indication : Paroxysmal nocturnal hemoglobinuria, atypical hemolytic uremic syndrome
Price 200000 INR / Bottle
Minimum Order Quantity : 1 Bottle
Dosage Form : Tablet
Salt Composition : Ivacaftor 150mg
Shelf Life : 24 months
Indication : Cystic Fibrosis in patients with specific gating (Class III) mutations in the CFTR gene
